Results 1 - 20 of 41,619 for All Library Resources
Sorted by:
Sorted by:
RelevanceOr hit Enter to replace sort method
- Relevance
- Date-newest
- Date-oldest
- Popularity
- Author
- Title
Show only
- Peer-reviewed Journals (35,836)
Refined by:
subject:
Medical Research
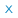
| Result Number | Material Type | Add to My Shelf Action | Record Details and Options |
|---|---|---|---|
| 1 |
Material Type: Book
|
|
Medical Research for Hire: The Political Economy of Pharmaceutical Clinical Trials2009 Jill A. Fisher ;ISBN: 0813545935 ;ISBN: 9780813545936 ;ISBN: 0813544092 ;ISBN: 9780813544090 ;ISBN: 0813544106 ;ISBN: 9780813544106 ;EISBN: 0813545935 ;EISBN: 9780813545936 ;DOI: 10.36019/9780813545936 ;OCLC: 301781578 ;LCCallNum: RM301.27.F57 2009Full text available |
| 2 |
Material Type: Article
|
|
Adaptive Designs for Clinical TrialsThe New England journal of medicine, 2016-07, Vol.375 (1), p.65-74 [Peer Reviewed Journal]Copyright © 2016 Massachusetts Medical Society. All rights reserved. ;ISSN: 0028-4793 ;EISSN: 1533-4406 ;DOI: 10.1056/NEJMra1510061 ;PMID: 27406349Full text available |
| 3 |
Material Type: Article
|
|
Validation of Time in Range as an Outcome Measure for Diabetes Clinical TrialsDiabetes care, 2019-03, Vol.42 (3), p.400-405 [Peer Reviewed Journal]2018 by the American Diabetes Association. ;Copyright American Diabetes Association Mar 1, 2019 ;2018 by the American Diabetes Association. 2018 ;ISSN: 0149-5992 ;EISSN: 1935-5548 ;DOI: 10.2337/dc18-1444 ;PMID: 30352896Full text available |
| 4 |
Material Type: Article
|
|
Criteria for the use of omics-based predictors in clinical trialsNature (London), 2013-10, Vol.502 (7471), p.317-320 [Peer Reviewed Journal]COPYRIGHT 2013 Nature Publishing Group ;COPYRIGHT 2013 Nature Publishing Group ;Copyright Nature Publishing Group Oct 17, 2013 ;The Author(s) 2013 ;ISSN: 0028-0836 ;EISSN: 1476-4687 ;DOI: 10.1038/nature12564 ;PMID: 24132288 ;CODEN: NATUASFull text available |
| 5 |
Material Type: Article
|
|
Revised STandards for Reporting Interventions in Clinical Trials of Acupuncture (STRICTA): extending the CONSORT statementPLoS medicine, 2010-06, Vol.7 (6), p.e1000261-e1000261 [Peer Reviewed Journal]MacPherson et al. 2010 ;2010 MacPherson et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited: MacPherson H, Altman DG, Hammerschlag R, Youping L, Taixiang W, et al. (2010) Revised STandards for Reporting Interventions in Clinical Trials of Acupuncture (STRICTA): Extending the CONSORT Statement. PLoS Med 7(6): e1000261. doi:10.1371/journal.pmed.1000261 ;ISSN: 1549-1676 ;ISSN: 1549-1277 ;EISSN: 1549-1676 ;DOI: 10.1371/journal.pmed.1000261 ;PMID: 20543992Full text available |
| 6 |
Material Type: Article
|
|
The COVID-19 pandemic and new clinical trial activationsCurrent controlled trials in cardiovascular medicine, 2021-04, Vol.22 (1), p.260-260, Article 260 [Peer Reviewed Journal]COPYRIGHT 2021 BioMed Central Ltd. ;COPYRIGHT 2021 BioMed Central Ltd. ;The Author(s) 2021. This work is published under http://creativecommons.org/licenses/by/4.0/ (the “License”). Notwithstanding the ProQuest Terms and Conditions, you may use this content in accordance with the terms of the License. ;The Author(s) 2021 ;ISSN: 1745-6215 ;EISSN: 1745-6215 ;EISSN: 1468-6694 ;DOI: 10.1186/s13063-021-05219-3 ;PMID: 33832534Full text available |
| 7 |
Material Type: Article
|
|
The impact of patient-reported outcome (PRO) data from clinical trials: a systematic review and critical analysisHealth and quality of life outcomes, 2019-10, Vol.17 (1), p.156-156, Article 156 [Peer Reviewed Journal]COPYRIGHT 2019 BioMed Central Ltd. ;2019. This work is licensed under http://creativecommons.org/licenses/by/4.0/ (the “License”). Notwithstanding the ProQuest Terms and Conditions, you may use this content in accordance with the terms of the License. ;The Author(s). 2019 ;ISSN: 1477-7525 ;EISSN: 1477-7525 ;DOI: 10.1186/s12955-019-1220-z ;PMID: 31619266Full text available |
| 8 |
Material Type: Article
|
|
2017 Cardiovascular and Stroke Endpoint Definitions for Clinical TrialsJournal of the American College of Cardiology, 2018-03, Vol.71 (9), p.1021-1034 [Peer Reviewed Journal]2018 American College of Cardiology Foundation and American Heart Association, Inc. ;Copyright © 2018 American College of Cardiology Foundation and American Heart Association, Inc. Published by Elsevier Inc. All rights reserved. ;Copyright Elsevier Limited Mar 6, 2018 ;ISSN: 0735-1097 ;EISSN: 1558-3597 ;DOI: 10.1016/j.jacc.2017.12.048 ;PMID: 29495982Full text available |
| 9 |
Material Type: Article
|
|
Cardiac MRI Endpoints in Myocardial Infarction Experimental and Clinical Trials: JACC Scientific Expert PanelJournal of the American College of Cardiology, 2019-07, Vol.74 (2), p.238-256 [Peer Reviewed Journal]Copyright © 2019 The Authors. Published by Elsevier Inc. All rights reserved. ;2019. The Authors ;ISSN: 0735-1097 ;EISSN: 1558-3597 ;DOI: 10.1016/j.jacc.2019.05.024 ;PMID: 31296297Full text available |
| 10 |
Material Type: Article
|
|
Impact of COVID-19 on oncology clinical trialsNature reviews. Drug discovery, 2020-06, Vol.19 (6), p.376-377 [Peer Reviewed Journal]COPYRIGHT 2020 Nature Publishing Group ;Nature 2020. ;ISSN: 1474-1776 ;EISSN: 1474-1784 ;DOI: 10.1038/d41573-020-00093-1 ;PMID: 32424343Full text available |
| 11 |
Material Type: Article
|
|
Risk-adapted monitoring is not inferior to extensive on-site monitoring: Results of the ADAMON cluster-randomised studyClinical trials (London, England), 2017-12, Vol.14 (6), p.584-596 [Peer Reviewed Journal]The Author(s) 2017 ;ISSN: 1740-7745 ;EISSN: 1740-7753 ;DOI: 10.1177/1740774517724165Digital Resources/Online E-Resources |
| 12 |
Material Type: Article
|
|
The COMET Handbook: version 1.0Current controlled trials in cardiovascular medicine, 2017-06, Vol.18 (Suppl 3), p.280-280, Article 280 [Peer Reviewed Journal]COPYRIGHT 2017 BioMed Central Ltd. ;The Author(s). 2017. This work is published under http://creativecommons.org/licenses/by/4.0/ (the “License”). Notwithstanding the ProQuest Terms and Conditions, you may use this content in accordance with the terms of the License. ;The Author(s). 2017 ;ISSN: 1745-6215 ;EISSN: 1745-6215 ;EISSN: 1468-6694 ;DOI: 10.1186/s13063-017-1978-4 ;PMID: 28681707Full text available |
| 13 |
Material Type: Article
|
|
Unsuccessful trial accrual and human subjects protections: An empirical analysis of recently closed trialsClinical trials (London, England), 2015-02, Vol.12 (1), p.77-83 [Peer Reviewed Journal]The Author(s) 2014 ;The Author(s) 2014. ;SAGE Publications © Feb 2015 ;ISSN: 1740-7745 ;EISSN: 1740-7753 ;DOI: 10.1177/1740774514558307 ;PMID: 25475878Full text available |
| 14 |
Material Type: Book
|
|
Clinical trials with missing data: a guide for practitioners (Statistics in practice).ISBN: 9781118762509 ;ISBN: 1118762509 ;ISBN: 9781118460702 ;ISBN: 1118460707 ;ISBN: 1118762533 ;ISBN: 9781118762530 ;ISBN: 1118762517 ;ISBN: 9781118762516 ;EISBN: 9781118762509 ;EISBN: 1118762509 ;EISBN: 1118762533 ;EISBN: 9781118762530 ;DOI: 10.1002/9781118762516 ;OCLC: 870951046Full text available |
| 15 |
Material Type: Article
|
|
Comparative safety of immune checkpoint inhibitors in cancer: systematic review and network meta-analysisBMJ (Online), 2018-11, Vol.363, p.k4226 [Peer Reviewed Journal]Published by the BMJ Publishing Group Limited. For permission to use (where not already granted under a licence) please go to http://group.bmj.com/group/rights-licensing/permissions. ;Published by the BMJ Publishing Group Limited. For permission to use (where not already granted under a licence) please go to http://group.bmj.com/group/rights-licensing/permissions 2018 BMJ This is an Open Access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/ . Notwithstanding the ProQuest Terms and Conditions, you may use this content in accordance with the terms of the License. ;Published by the BMJ Publishing Group Limited. For permission to use (where not already granted under a licence) please go to 2018 BMJ ;ISSN: 0959-8138 ;EISSN: 1756-1833 ;DOI: 10.1136/bmj.k4226 ;PMID: 30409774Full text available |
| 16 |
Material Type: Article
|
|
Patient-centric trials for therapeutic development in precision oncologyNature (London), 2015-10, Vol.526 (7573), p.361-370 [Peer Reviewed Journal]COPYRIGHT 2015 Nature Publishing Group ;COPYRIGHT 2015 Nature Publishing Group ;Copyright Nature Publishing Group Oct 15, 2015 ;ISSN: 0028-0836 ;EISSN: 1476-4687 ;DOI: 10.1038/nature15819 ;PMID: 26469047 ;CODEN: NATUASFull text available |
| 17 |
Material Type: Article
|
|
It is unprecedented: trial management during the COVID-19 pandemic and beyondCurrent controlled trials in cardiovascular medicine, 2020-09, Vol.21 (1), p.784-784, Article 784 [Peer Reviewed Journal]COPYRIGHT 2020 BioMed Central Ltd. ;The Author(s) 2020. This work is published under http://creativecommons.org/licenses/by/4.0/ (the “License”). Notwithstanding the ProQuest Terms and Conditions, you may use this content in accordance with the terms of the License. ;The Author(s) 2020 ;ISSN: 1745-6215 ;EISSN: 1745-6215 ;EISSN: 1468-6694 ;DOI: 10.1186/s13063-020-04711-6 ;PMID: 32917258Full text available |
| 18 |
Material Type: Article
|
|
Three randomized controlled trials evaluating the impact of "spin" in health news stories reporting studies of pharmacologic treatments on patients'/caregivers' interpretation of treatment benefitBMC medicine, 2019-06, Vol.17 (1), p.105-105, Article 105 [Peer Reviewed Journal]COPYRIGHT 2019 BioMed Central Ltd. ;COPYRIGHT 2019 BioMed Central Ltd. ;2019. This work is licensed under http://creativecommons.org/licenses/by/4.0/ (the “License”). Notwithstanding the ProQuest Terms and Conditions, you may use this content in accordance with the terms of the License. ;Distributed under a Creative Commons Attribution 4.0 International License ;The Author(s). 2019 ;ISSN: 1741-7015 ;EISSN: 1741-7015 ;DOI: 10.1186/s12916-019-1330-9 ;PMID: 31159786Full text available |
| 19 |
Material Type: Article
|
|
A limited number of medicines pragmatic trials had potential for waived informed consent following the 2016 CIOMS ethical guidelinesJournal of clinical epidemiology, 2019-10, Vol.114, p.60-71 [Peer Reviewed Journal]2019 Elsevier Inc. ;Copyright © 2019 Elsevier Inc. All rights reserved. ;2019. Elsevier Inc. ;ISSN: 0895-4356 ;ISSN: 1878-5921 ;EISSN: 1878-5921 ;DOI: 10.1016/j.jclinepi.2019.06.007 ;PMID: 31212001Full text available |
| 20 |
Material Type: Article
|
|
Many-to-one comparisons after safety selection in multi-arm clinical trialsPloS one, 2017-06, Vol.12 (6), p.e0180131-e0180131 [Peer Reviewed Journal]COPYRIGHT 2017 Public Library of Science ;COPYRIGHT 2017 Public Library of Science ;2017 Hlavin et al. This is an open access article distributed under the terms of the Creative Commons Attribution License: http://creativecommons.org/licenses/by/4.0/ (the “License”), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Notwithstanding the ProQuest Terms and Conditions, you may use this content in accordance with the terms of the License. ;2017 Hlavin et al 2017 Hlavin et al ;ISSN: 1932-6203 ;EISSN: 1932-6203 ;DOI: 10.1371/journal.pone.0180131 ;PMID: 28651023Full text available |
Personalize your results
Refine Search Results
Expand My Results
Show only
- Peer-reviewed Journals (35,836)
Refine My Results
Creation Date
- Before 1969 (27)
- 1969 To 1982 (96)
- 1983 To 1996 (861)
- 1997 To 2011 (6,880)
- After 2011 (33,849)
-
More options

New Records
- New From Last Week (4)
- New From Last Month (130)
- New From Last 3 Month (584)
-
More options

Resource Type
- Articles (38,488)
- magazinearticle (1,226)
- Newspaper Articles (569)
- Books (539)
- Book Chapters (454)
- Reports (125)
- Newsletter Articles (110)
- Reviews (90)
- Conference Proceedings (8)
- References (5)
- Text Resources (2)
- Thesises (postgraduate) (2)
- Web Resources (1)
-
More options

Subject
- Clinical Trials (38,633)
- Science & Technology (31,367)
- Life Sciences & Biomedicine (26,157)
- Humans (20,052)
- Medicine, Experimental (12,735)
- Patients (11,929)
- Female (7,859)
- Care And Treatment (7,222)
- Male (6,660)
- Analysis (6,349)
- Health Aspects (6,228)
- Cancer (5,915)
- Studies (5,857)
- Medicine (5,630)
- Science & Technology - Other Topics (5,019)
- Multidisciplinary Sciences (4,920)
- Oncology (4,624)
- Middle Aged (4,604)
- Adult (4,560)
-
More options

Databases
- ProQuest Central (37,367)
- PubMed Central (23,679)
- DOAJ Directory of Open Access Journals (22,783)
- MEDLINE (21,174)
- GFMER Free Medical Journals (21,022)
- ROAD: Directory of Open Access Scholarly Resources (20,645)
- Springer Nature OA Free Journals (8,322)
- Public Library of Science (PLoS) (4,762)
- ProQuest One Psychology (4,370)
- HighWire Press (Free Journals) (3,177)
- Coronavirus Research Database (2,413)
- Taylor & Francis Open Access (1,707)
- DOVE Medical Press Journals (1,642)
- Wiley Online Library Open Access (1,429)
- Medknow Open Access Medical Journals (710)
- Journals@Ovid Open Access Journal Collection Rolling (631)
- BMJ Open Access Journals (585)
- SWEPUB Freely available online (537)
- Free E- Journals (472)
- Nature Open Access (461)
-
More options

Language
Journal Title
- Bmc Medical Research Methodology (395)
- Bmj (807)
- Cancers (409)
- Current Controlled Trials In Cardiovascular Medicine (1,269)
- Nature (473)
- New York Times (398)
- Plos One (4,311)
- The Lancet (1,000)
- The New England Journal Of Medicine (430)
- Trials (1,346)
-
More options





